In Vitro Biocompatibility
In vitro biocompatibility testing plays a critical role in the development of modern medical devices and healthcare materials. Any material that comes into contact with the human body must be carefully evaluated to ensure that it does not cause harmful biological reactions. Whether the product is a surgical instrument, implantable device, pharmaceutical packaging material, or wearable health technology, its interaction with living cells and tissues must be fully understood before it can be safely introduced to the market.
By conducting in vitro studies in a controlled laboratory environment, researchers can evaluate how cells respond when exposed to a particular material. These tests help determine whether a material causes toxicity, irritation, inflammation, or other adverse biological responses. This process is essential for identifying potential risks early in product development, allowing manufacturers to refine their materials and designs before proceeding to more advanced testing or regulatory submission.
Why Choose Genuine Testing for In Vitro Biocompatibility?
Regulatory Compliance
We follow ISO 10993 standards and other global guidelines to help you meet regulatory approval for your medical devices.Experienced Team
Our scientists bring deep expertise in biomedical materials and testing methods.Cost-Effective & Ethical
In vitro methods are faster, more affordable, and more humane than traditional in vivo testing.Clear, Actionable Reports
You get detailed and easy-to-understand results to support your development process.

Our Testing Capabilities
Our in vitro tests help you determine the safety and suitability of your materials and products, including:
Cytotoxicity testing – Evaluating whether materials harm or kill cells in culture.
Irritation and sensitization potential – Assessing how materials might cause skin or mucosal irritation.
Hemocompatibility – Testing compatibility with blood and blood components.
Endotoxin testing – Detecting pyrogens and contaminants that may cause adverse reactions.
Material characterization – Analyzing composition to ensure consistency and compliance.
Applications & Industries
We support manufacturers and developers of:
Medical devices & implants
Surgical instruments
Pharmaceutical packaging
Tissue engineering materials
Wearable health technologies
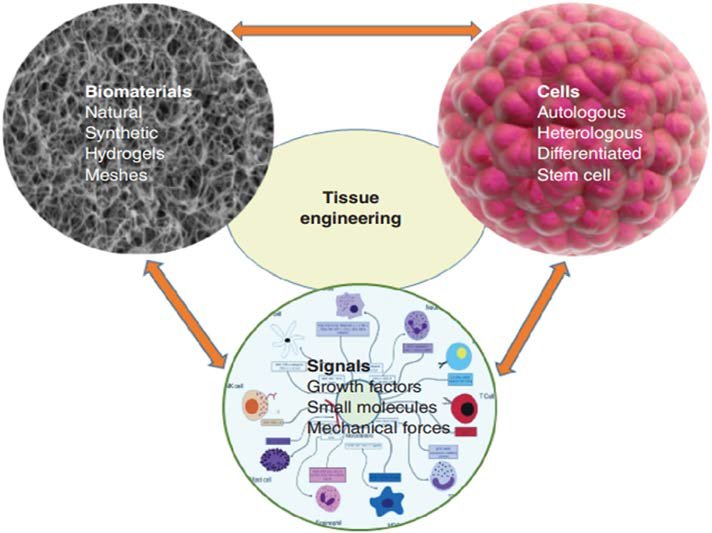
The Role of Biocompatibility in Medical Device Development
Biocompatibility is one of the most important factors in the development of any medical product that interacts with the human body. When materials are introduced into biological systems, they may trigger a range of cellular responses depending on their chemical composition, surface properties, and manufacturing processes. Even small changes in material formulation can influence how cells react, which is why careful testing is necessary throughout product development.
In vitro biocompatibility testing allows developers to evaluate these interactions at an early stage. Through carefully designed experiments, scientists expose cultured cells to materials or extracts derived from them and observe how the cells behave over time. These observations help identify whether the material affects cell viability, growth, or metabolic activity.
Such evaluations are particularly important for implantable devices that remain inside the body for extended periods. Long-term exposure to certain materials can potentially trigger inflammatory responses or interfere with normal tissue function. By studying cellular responses in vitro, developers can identify these issues early and modify materials accordingly. This proactive approach helps reduce costly redesigns later in the development cycle and accelerates the pathway toward regulatory approval.
The increasing complexity of modern medical devices also makes biocompatibility evaluation more important than ever. Advanced biomaterials, coatings, polymers, and composite materials are now commonly used in devices ranging from orthopedic implants to drug delivery systems. Each of these materials must undergo thorough safety evaluation to ensure that they perform as intended without compromising patient health.

Regulatory Standards and Global Compliance
Compliance with international regulatory standards is essential for any organization seeking to commercialize medical products. Regulatory authorities around the world require manufacturers to demonstrate the safety of their materials through scientifically validated testing methods. Among the most widely recognized frameworks for biological safety evaluation is the ISO 10993 series of standards, which provides comprehensive guidance for assessing the biocompatibility of medical devices.
These standards outline a risk-based approach that considers the type of device, the duration of contact with the body, and the nature of the tissues involved. Based on these factors, specific tests may be required to evaluate cytotoxicity, irritation potential, sensitization, systemic toxicity, and other biological effects. By following this framework, manufacturers can generate data that supports regulatory submissions across multiple global markets.
At Genuine Testing, we conduct in vitro biocompatibility studies in accordance with internationally recognized guidelines to ensure that our results are accepted by regulatory bodies worldwide. Our laboratory procedures are designed to maintain strict quality control, from sample preparation and cell culture conditions to analytical measurements and data reporting.
Advanced Laboratory Techniques and Scientific Expertise
The accuracy and reliability of in vitro biocompatibility testing depend heavily on the quality of laboratory techniques and the expertise of the scientific team conducting the studies. Modern testing laboratories use advanced equipment and carefully controlled experimental procedures to ensure that results are both reproducible and scientifically valid.
Cell culture technology lies at the heart of most in vitro biocompatibility assessments. Researchers grow living cells under controlled conditions and expose them to the test materials or extracts derived from those materials. By monitoring how these cells respond, scientists can identify potential toxic effects and measure changes in cellular metabolism, morphology, or viability.
In addition to cell-based assays, advanced analytical techniques are often used to evaluate the chemical composition of materials and detect potential contaminants. These analyses help ensure that materials meet strict quality requirements and remain consistent across production batches. The combination of biological testing and chemical characterization provides a comprehensive understanding of how materials interact with biological systems.

Supporting Innovation Across Healthcare Industries
As healthcare technologies continue to evolve, the demand for reliable biocompatibility testing services continues to grow. Emerging innovations such as wearable medical devices, tissue-engineered materials, and advanced drug delivery systems all require careful safety evaluation before they can reach patients. Each of these technologies involves materials that interact with the human body in unique ways, making thorough testing essential.
Wearable health technologies, for example, often involve prolonged skin contact. Even though these devices may appear simple, the materials used in their construction must be carefully evaluated to ensure they do not cause irritation or allergic reactions during extended use. In vitro testing helps manufacturers identify suitable materials that remain comfortable and safe for everyday wear.
Similarly, tissue engineering and regenerative medicine rely on biomaterials designed to support cell growth and tissue regeneration. These materials must provide an environment that encourages healthy cellular behavior without triggering harmful responses. Biocompatibility testing ensures that such materials are suitable for their intended biological applications.
