Biomaterials & Medical-Grade Materials
Advanced Testing for Biomaterials & Medical-Grade Performance
Biomaterials and medical-grade materials are engineered to function safely and reliably within or in contact with the human body. These materials are used in orthopedic implants, cardiovascular devices, dental systems, surgical instruments, prosthetics, and advanced medical equipment. Because of their direct impact on patient health and safety, biomaterials must meet rigorous mechanical, structural, and durability standards while complying with strict regulatory frameworks. At Genuine Testing, we provide comprehensive biomaterials testing and medical-grade materials evaluation services designed to validate safety, strength, durability, and long-term performance. As an advanced materials testing laboratory and Contract Research Organization (CRO), we support medical device manufacturers, research institutions, and healthcare innovators with accurate, defensible, and regulatory-ready data.
Medical-grade materials must withstand complex loading conditions, repeated mechanical stress, sterilization processes, and long-term exposure to biological environments. Whether evaluating titanium orthopedic implants, medical-grade polymers, bioactive ceramics, or implantable composite materials, our laboratory ensures that products meet the highest standards of performance and reliability. With rapid growth in minimally invasive devices, 3D-printed implants, and next-generation biomaterials, robust material validation is more critical than ever. Our testing programs help reduce development risk, accelerate regulatory approval, and ensure long-term clinical success.
Mechanical Performance & Structural Integrity of Implant Materials
Implantable and load-bearing medical devices must maintain structural integrity under dynamic physiological conditions. Orthopedic implants, spinal fixation systems, joint replacements, and dental implants are subjected to cyclic loading, compressive forces, and torsional stresses throughout their service life. Even minor weaknesses can lead to premature failure and serious clinical consequences.
Our biomaterials testing laboratory performs detailed mechanical evaluation to determine tensile strength, fatigue resistance, compression behavior, and fracture characteristics. Fatigue testing is particularly important for orthopedic implant materials such as titanium alloys and cobalt-chromium systems, which must endure millions of loading cycles without degradation. Medical-grade polymers used in catheters, surgical instruments, and implantable components require validation of flexibility, resilience, and stress resistance. Mechanical testing ensures that these materials maintain performance under repeated use or long-term implantation.

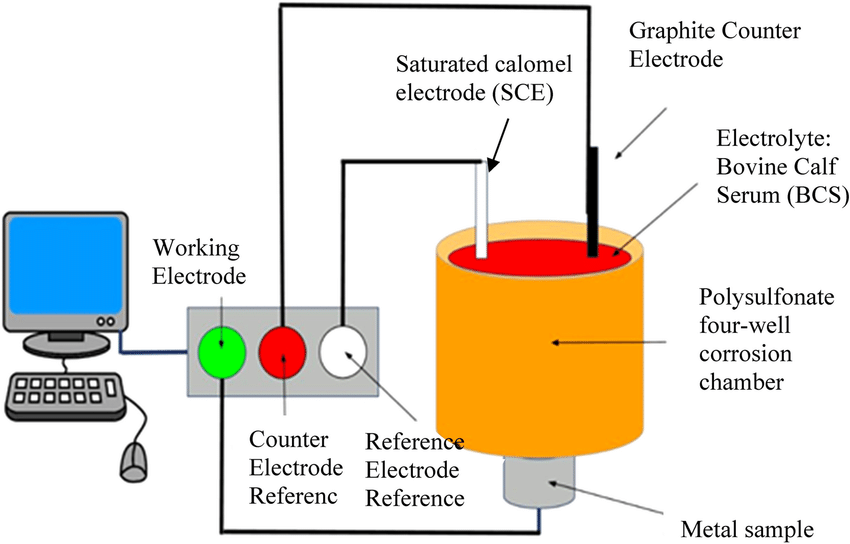
Durability, Corrosion & Environmental Resistance
Medical-grade materials must maintain performance in complex biological and sterilization environments. Exposure to body fluids, varying pH levels, temperature fluctuations, and repeated sterilization cycles can significantly affect material behavior over time.
Corrosion resistance testing is essential for metallic implants and surgical instruments. Titanium and stainless steel components must resist degradation and surface breakdown that could compromise mechanical integrity or release unwanted particles. Our evaluation programs simulate physiological conditions to assess corrosion performance and long-term durability. Medical polymers and elastomeric biomaterials are also sensitive to environmental factors. Heat, moisture, and sterilization processes such as autoclaving or gamma irradiation can alter mechanical properties and surface characteristics. Through controlled environmental testing, we evaluate property retention and dimensional stability after exposure to sterilization cycles.
Surface characterization plays a crucial role in biomaterials validation. Surface roughness, microstructural integrity, and coating performance can influence mechanical stability and long-term functionality. By integrating surface analysis with mechanical testing, we provide a comprehensive assessment of implant and device material reliability.
As medical devices become increasingly miniaturized and precision-driven, understanding how materials behave under combined mechanical and environmental stresses is vital. Our laboratory ensures that biomaterials meet durability requirements before entering clinical use.

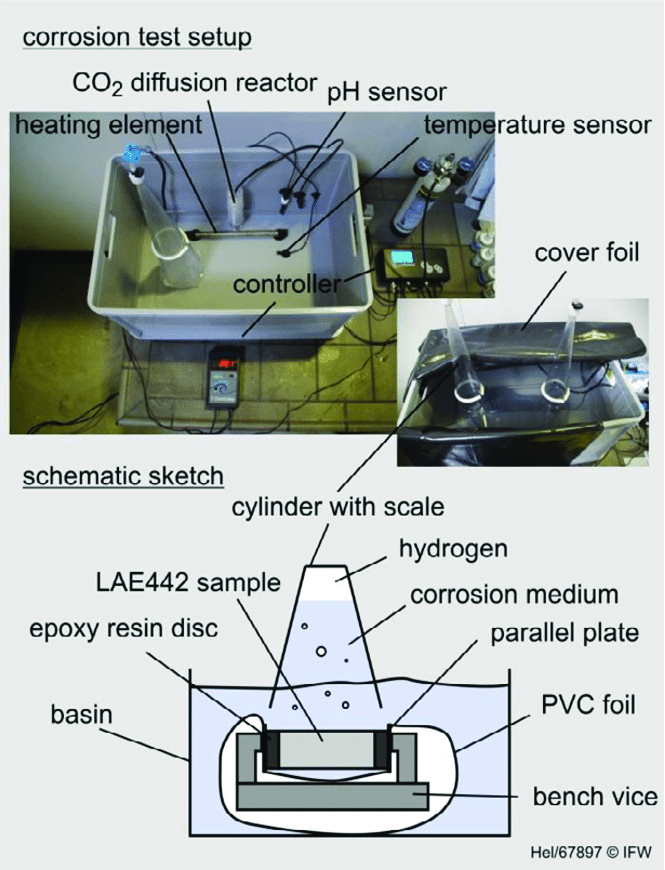
Medical-Grade Polymers, Ceramics & Advanced Biomaterials
The biomaterials sector continues to evolve rapidly, with innovations in polymer chemistry, bioactive ceramics, and composite implant materials. Modern medical devices incorporate advanced polymers designed for flexibility, biostability, and performance under long-term physiological conditions.
Medical-grade polymer testing includes evaluation of tensile properties, creep resistance, and resistance to environmental degradation. Flexible materials used in catheters, tubing, and minimally invasive devices must retain mechanical performance while remaining safe for patient contact. Bioactive ceramics and glass-based materials used in dental implants and bone graft applications require structural and mechanical validation. These materials must provide sufficient strength while supporting biological integration. Mechanical characterization ensures that they meet structural requirements without compromising function.
Additive manufacturing has introduced new possibilities in patient-specific implants and surgical guides. 3D-printed biomaterials often exhibit unique microstructures and anisotropic properties that require specialized validation. Our testing programs address these complexities to ensure that innovative manufacturing approaches do not compromise reliability.
By evaluating traditional and emerging biomaterials with equal rigor, we support innovation while maintaining strict quality assurance.


Regulatory Compliance & Risk Mitigation
Medical device manufacturers operate within highly regulated environments where material validation plays a central role in regulatory submissions and product approvals. Comprehensive biomaterials testing provides the technical documentation required to demonstrate safety, performance, and reliability.
Our laboratory follows internationally recognized testing methodologies to ensure data integrity and regulatory readiness. Accurate mechanical testing, durability studies, and failure analysis contribute to smoother approval pathways and reduced time to market. Risk mitigation is another critical component of biomaterials evaluation. Early identification of mechanical weaknesses, corrosion susceptibility, or environmental sensitivity prevents costly recalls and protects brand reputation. By implementing rigorous validation programs during product development, manufacturers can reduce uncertainty and strengthen clinical confidence.
Collaborative Support for Medical Innovation
At Genuine Testing, we understand that medical-grade materials testing is not simply about compliance; it is about safeguarding patient outcomes and enabling innovation. Our team collaborates closely with R&D engineers, regulatory specialists, and quality assurance teams to design testing programs aligned with specific device applications.
We provide clear, comprehensive reporting that translates complex data into actionable insights. Whether supporting early-stage research, design validation, or post-market investigation, our CRO-level expertise ensures confidentiality, accuracy, and technical excellence. As the medical industry continues to advance toward personalized medicine, minimally invasive procedures, and smart implant technologies, the demand for robust biomaterials validation will continue to grow. Our laboratory remains committed to supporting these advancements with reliable, high-precision testing services.

Another critical consideration in medical-grade materials testing is manufacturing consistency. Variations in processing methods, surface treatments, additive manufacturing parameters, or heat treatments can significantly affect microstructure and mechanical behavior. By conducting batch-to-batch material validation and comparative performance analysis, we ensure consistent quality and reproducibility across production cycles. This level of scrutiny is particularly important for implantable components and precision medical instruments where dimensional stability and structural integrity are non-negotiable.
Emerging technologies such as bioresorbable materials, smart implants, and hybrid composite biomaterials further increase the need for specialized testing expertise. These advanced materials are engineered to interact dynamically with biological systems, and their degradation rates or structural responses must be carefully characterized. Comprehensive materials evaluation supports innovation while maintaining strict safety margins. At Genuine Testing, we combine scientific rigor with industry insight to deliver data-driven validation that strengthens product performance, regulatory confidence, and long-term clinical reliability.
