Biomedical Analysis
Biomedical materials must meet the highest standards of safety, reliability, and performance. Whether used in implantable devices, surgical instruments, diagnostic systems, drug delivery platforms, or wearable health technologies, these materials operate in complex biological environments where mechanical integrity, chemical stability, and biocompatibility are critical. Genuine Testing provides comprehensive biomedical analysis services to support research and development, regulatory submission, quality assurance, and failure investigation.
As an independent materials testing laboratory and Contract Research Organization (CRO), we deliver objective, high-precision data that helps manufacturers and research teams ensure compliance, enhance performance, and protect patient safety. Our analytical capabilities support metals, polymers, ceramics, coatings, composites, and advanced functional materials used in medical and life science applications.
Why Choose Genuine Testing for Biomedical Analysis?
Precision Insights into Biomedical Materials
We deliver accurate, detailed information on biomaterials, coatings, interfaces, and contaminants to ensure your products meet regulatory and functional requirements.
Expertise Across Applications
From implantable devices and prosthetics to drug delivery systems and diagnostic equipment, our services support a wide range of biomedical innovations.
Advanced Analytical Techniques
We use state-of-the-art tools and methodologies to investigate structure, chemistry, and performance — helping you improve product reliability and biocompatibility.
Fast & Reliable Results
Our efficient processes and knowledgeable team ensure timely, actionable data to accelerate your projects.


Our Biomedical Testing Capabilities
We offer a comprehensive suite of analytical services for biomedical materials and devices, including:
- Structural and surface analysis of biomaterials (e.g., polymers, ceramics, metals)
- Microstructure and morphology assessment of medical devices
- Chemical composition and contamination analysis
- Coating thickness and uniformity measurement
- Biocompatibility-related surface chemistry studies
Commonly used techniques:
✅ Scanning Electron Microscopy (SEM)
✅ Transmission Electron Microscopy (TEM)
✅ Energy Dispersive X-Ray Spectroscopy (EDS)
✅ X-Ray Photoelectron Spectroscopy (XPS)
✅ Fourier Transform Infrared Spectroscopy (FTIR)
✅ Time-of-Flight Secondary Ion Mass Spectrometry (TOF-SIMS)
Industries We Serve
Our biomedical analysis services are trusted by clients in diverse sectors:
- Medical Device Manufacturers — Assuring material integrity, cleanliness, and regulatory compliance.
- Pharmaceutical & Biotech — Supporting drug delivery systems and biocompatibility studies.
- Academic & Clinical Research — Enabling breakthroughs in medical materials and technologies.
- Regulatory & QA Teams — Providing data to meet stringent international standards.
We work closely with you at every stage of your product development lifecycle to deliver insights that matter.

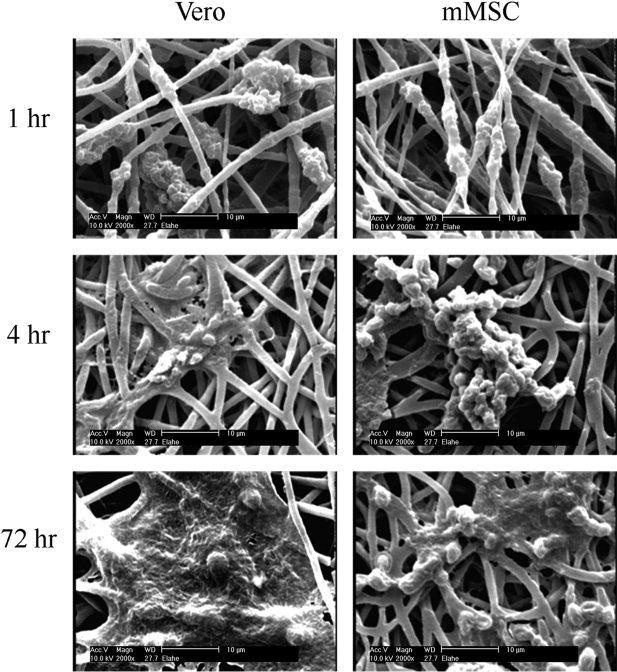
Biomaterials Characterization & Microstructural Evaluation
The structural integrity of biomaterials directly influences long-term performance within the human body. Grain size distribution in metallic implants, porosity in bone scaffolds, polymer morphology in catheters, and coating adhesion on orthopedic components all affect durability and biological response.
Our laboratory performs detailed microstructural analysis using optical microscopy, scanning electron microscopy, and advanced imaging techniques to assess surface morphology, internal structure, and material uniformity. Cross-sectional analysis enables evaluation of layered systems, including coated implants, stents, and drug-eluting devices. High-resolution imaging provides insight into defects such as voids, inclusions, microcracks, and delamination that may compromise device integrity.
Through precise characterization, we help manufacturers validate design specifications and ensure consistent production quality.
Surface Chemistry & Interface Analysis
Surface properties are particularly critical in biomedical applications because the interface between material and tissue governs biological response. Surface roughness, chemical composition, coating thickness, and cleanliness influence osseointegration, cell adhesion, antimicrobial performance, and corrosion resistance.
Our advanced surface analysis services evaluate chemical composition, coating uniformity, and potential contamination. Elemental mapping and compositional testing detect trace residues, manufacturing byproducts, or degradation products that may impact biocompatibility. We also assess surface treatments such as passivation layers, plasma coatings, and antimicrobial finishes to ensure stability and functional performance.
Comprehensive surface characterization supports both product optimization and regulatory documentation requirements.
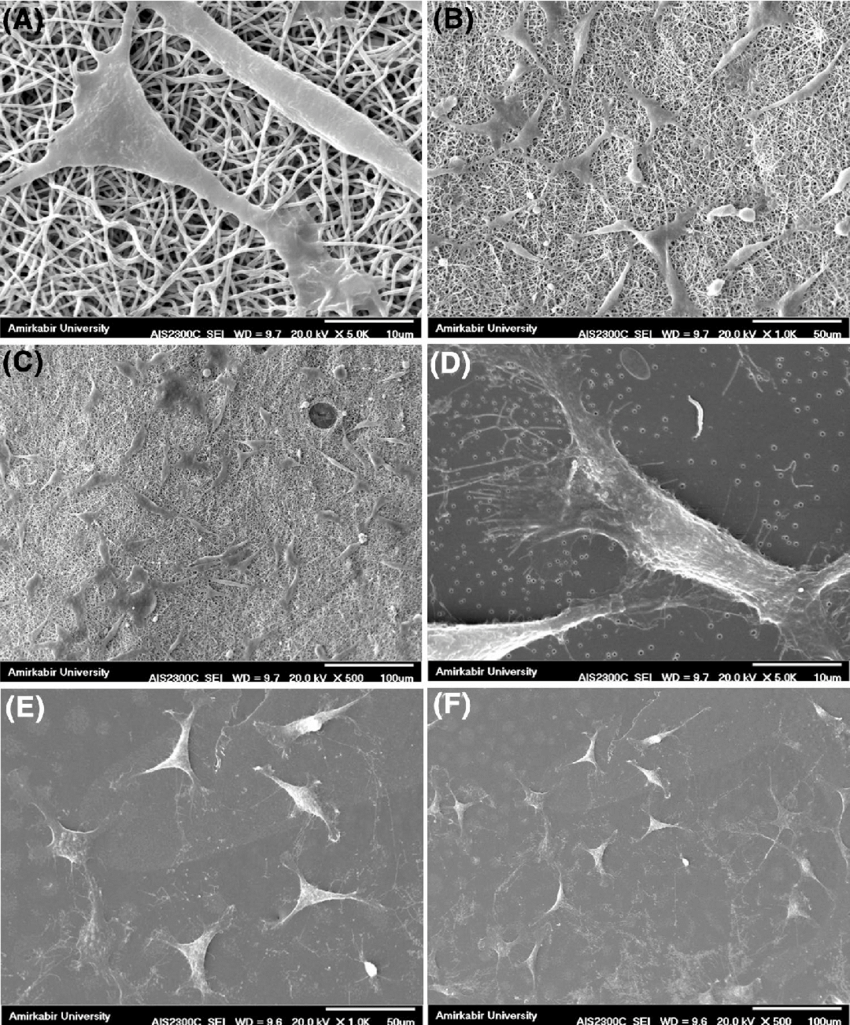
Medical Device Failure Analysis
When medical devices fail—whether through fracture, corrosion, wear, or material degradation—thorough investigation is essential. Biomedical failure analysis requires careful handling, precise imaging, and expert interpretation to determine root causes while preserving evidence.
Our laboratory conducts detailed fractography, corrosion analysis, and compositional evaluation to identify crack initiation sites, fatigue patterns, environmental damage, and manufacturing defects. We examine retrieved implants, surgical components, and device fragments to determine whether failure resulted from material selection, processing irregularities, mechanical overload, or in-service degradation.
Clear, defensible reporting provides valuable insight for corrective actions, product redesign, quality system improvements, and legal documentation.
Biocompatibility Support & Regulatory Alignment
Biomedical products must comply with stringent regulatory standards governing material safety and performance. While biocompatibility testing often involves biological assays, materials characterization forms a critical foundation for regulatory submissions and risk assessments.
We provide analytical data that supports biocompatibility evaluations, including surface chemistry verification, contamination analysis, degradation studies, and extractables assessment. Our documentation aligns with industry-recognized standards and regulatory frameworks to facilitate smooth submission processes.
Independent third-party testing enhances credibility with regulatory authorities, notified bodies, and healthcare partners by ensuring transparency and traceability throughout product development.

Wear, Corrosion & Degradation Studies
Biomedical materials are continuously exposed to mechanical stress, bodily fluids, and biochemical interactions. Over time, these factors can cause wear, corrosion, ion release, or structural weakening.
Our laboratory conducts simulated environmental testing and post-exposure analysis to evaluate corrosion resistance, coating durability, and wear behavior. Microscopic examination of exposed samples reveals surface pitting, tribological damage, and microstructural changes. These findings help predict service life and ensure long-term safety for implantable and external medical devices.
Understanding degradation pathways supports safer product design and improved material selection.
Polymer & Soft Material Analysis in Healthcare
Polymers and elastomers are widely used in catheters, tubing, syringes, packaging, prosthetics, and drug delivery systems. Their flexibility, chemical stability, and processability make them indispensable in modern medicine, yet they must maintain performance under sterilization, storage, and physiological conditions.
We analyze polymer morphology, thermal stability, additive dispersion, and degradation behavior using advanced analytical techniques. Surface and bulk characterization identifies potential leachables, plasticizer migration, or structural changes following sterilization processes such as gamma irradiation or autoclaving.
These evaluations ensure that medical-grade polymers retain mechanical integrity and safety throughout their intended lifecycle.


Supporting Innovation in Medical Technology
Biomedical innovation is rapidly advancing in areas such as minimally invasive surgery, bioresorbable implants, smart wearable devices, and regenerative medicine scaffolds. Each advancement introduces new material challenges that demand precise characterization and rigorous validation.
Genuine Testing partners with medical device manufacturers, research institutions, and healthcare technology developers to deliver comprehensive biomedical analysis tailored to each project’s objectives. Our integrated approach connects microstructural imaging, compositional analysis, thermal evaluation, and mechanical testing to provide a complete understanding of material behavior in medical environments.
Through scientific rigor, regulatory awareness, and independent verification, our Biomedical Analysis services help ensure that medical materials perform safely, reliably, and consistently—ultimately supporting improved patient outcomes and advancing the future of healthcare technology.
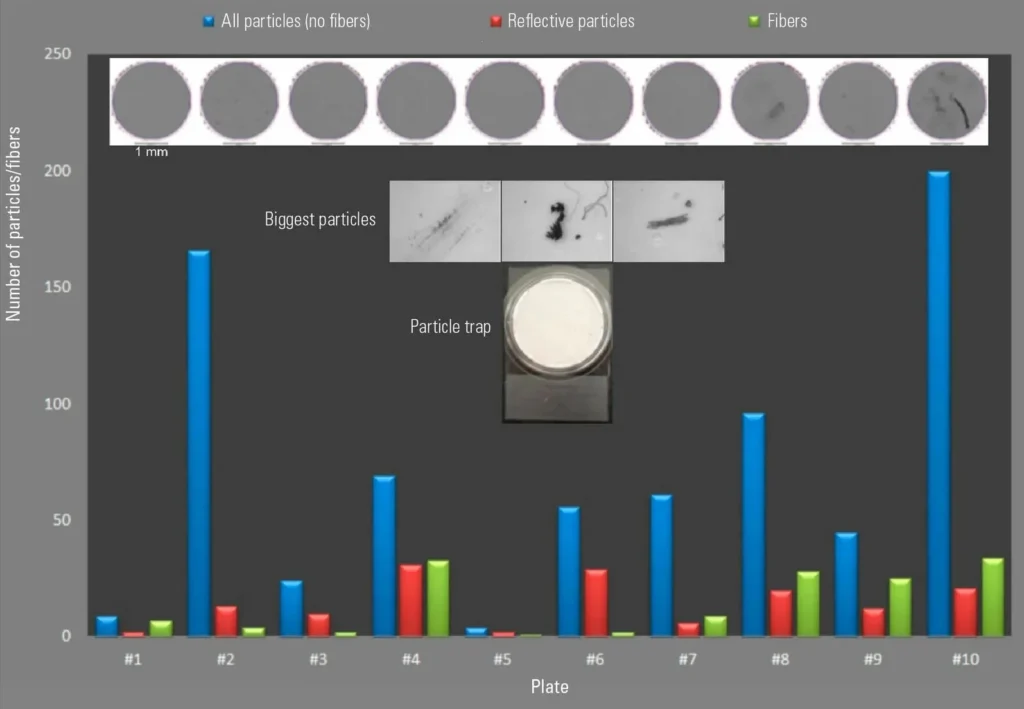
Extractables, Leachables & Chemical Stability Studies
Chemical stability is a critical component of biomedical safety. Materials used in implants, drug delivery systems, IV bags, syringes, and pharmaceutical packaging must not release harmful substances when exposed to bodily fluids, sterilization processes, or long-term storage conditions. Even trace-level extractables and leachables can influence toxicity profiles, drug efficacy, or patient outcomes.
Our biomedical analysis services include comprehensive extractables and leachables studies to identify potential chemical migrants originating from polymers, adhesives, coatings, elastomers, and composite systems. Using advanced analytical instrumentation, we evaluate volatile, semi-volatile, and non-volatile compounds under simulated clinical and storage environments. These studies help manufacturers assess chemical compatibility, ensure regulatory alignment, and reduce risk during product lifecycle management. By integrating chemical analysis with material characterization, we provide a clear understanding of how formulation choices and processing conditions influence long-term material stability.
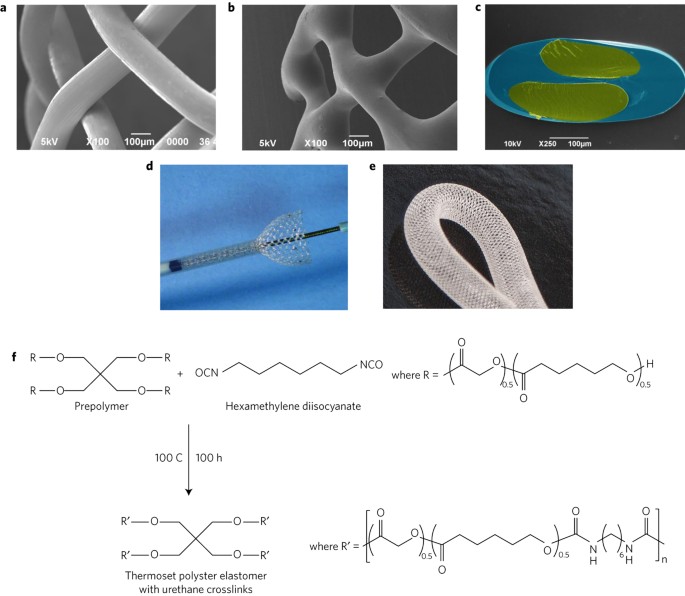
Sterilization Impact Assessment
Sterilization is essential for medical device safety, yet sterilization methods can significantly alter material properties. Gamma irradiation, ethylene oxide exposure, steam autoclaving, and plasma sterilization may induce polymer chain scission, oxidation, discoloration, embrittlement, or changes in surface chemistry.
We evaluate the impact of sterilization processes on mechanical strength, surface morphology, chemical composition, and microstructural integrity. Comparative pre- and post-sterilization analysis reveals subtle structural changes that may affect performance over time. These assessments are particularly important for implantable polymers, bioresorbable devices, and precision-engineered components where dimensional stability and mechanical reliability are critical.
Understanding sterilization-induced effects allows manufacturers to select optimal materials and validate sterilization protocols without compromising device longevity.
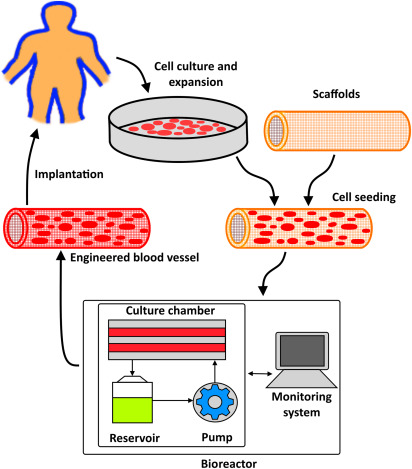
Bioresorbable & Regenerative Materials Evaluation
Bioresorbable implants and regenerative scaffolds represent a rapidly expanding area of biomedical innovation. These materials are designed to gradually degrade within the body while supporting tissue regeneration, eliminating the need for surgical removal.
Because controlled degradation is fundamental to their function, detailed analysis of structural evolution, porosity changes, and chemical breakdown is essential. Our laboratory evaluates degradation kinetics, microstructural integrity over time, and byproduct formation under simulated physiological conditions. Imaging and compositional analysis provide insight into how scaffold architecture supports cell growth while maintaining temporary mechanical strength.
Through precise monitoring of degradation pathways, we help ensure predictable performance and safe absorption profiles.

Wearable & Smart Medical Device Materials
The growth of wearable health technologies and smart medical devices introduces new material considerations, including flexibility, skin compatibility, electronic functionality, and environmental durability. These devices often combine polymers, conductive materials, thin films, and sensor components within compact, multilayered systems.
Our biomedical analysis services support the evaluation of flexible substrates, conductive coatings, microelectronic interfaces, and encapsulation materials. We examine adhesion between layers, surface roughness affecting sensor accuracy, and long-term stability under sweat exposure, mechanical flexing, and temperature fluctuations.
By correlating material performance with real-world usage conditions, we help ensure that wearable healthcare technologies remain reliable, comfortable, and safe for extended patient use.
