Extractables & Leachables
Extractables and leachables analysis plays a critical role in ensuring the safety, quality, and stability of pharmaceutical products, medical devices, and other sensitive materials. Many products are stored, transported, or delivered using containers, packaging materials, tubing, closures, or polymer-based components. While these materials are carefully selected for compatibility, they may still contain small quantities of additives, processing aids, residual monomers, stabilizers, plasticizers, or other chemical substances that can migrate into the product over time.
Extractables refer to compounds that can be released from a material when it is exposed to aggressive laboratory extraction conditions, such as elevated temperatures, strong solvents, or extended contact times. These studies are designed to identify the full range of potential chemical substances that could be present within a material.At Genuine Testing, we provide comprehensive Extractables and Leachables analysis to ensure the safety, quality, and compliance of materials that come into contact with drug products, food, or medical devices. Our Extractables and Leachables testing identifies and quantifies potentially harmful chemicals that may migrate from packaging, containers, or components into your product.
Why Choose Genuine Testing for E&L Testing?
Regulatory Expertise
We design and execute testing strategies that align with current regulatory guidelines and industry standards.Advanced Analytical Tools
Our laboratory is equipped with sensitive, state-of-the-art instrumentation for detecting even trace-level contaminants.Cross-Industry Experience
We work with pharmaceutical, medical device, food contact, and consumer product manufacturers.End-to-End Support
From method development and validation to data interpretation and reporting, we guide you through the entire process.
What Are Extractables & Leachables?
Extractables are compounds that can be extracted from materials under exaggerated conditions (e.g., high heat, solvents).
Leachables are compounds that actually migrate into the product under normal storage or use conditions.
Both can pose safety risks and impact product performance, making their identification and quantification critical for regulatory approval and product integrity.
Our Extractables & Leachables Testing Capabilities
We offer customized E&L testing programs, including:
Controlled extraction studies (extractables)
Simulation and real-time storage studies (leachables)
Identification and quantification of organic and inorganic compounds
Risk assessment and toxicological evaluation
Method development and validation
Techniques we employ:
Gas Chromatography-Mass Spectrometry (GC-MS)
Liquid Chromatography-Mass Spectrometry (LC-MS/MS)
Inductively Coupled Plasma Mass Spectrometry (ICP-MS)
Fourier Transform Infrared Spectroscopy (FTIR)
Headspace GC and Thermal Desorption

Applications & Industries
Our Extractables and Leachables analysis serves:
Pharmaceuticals & Biologics — primary and secondary packaging, container-closure systems
Medical Devices — catheters, tubing, syringes, implantable devices
Food Contact Materials — packaging films, containers, coatings
Cosmetics & Personal Care — packaging and applicators
The Importance of E&L Studies in Regulated Industries
In industries such as pharmaceuticals, biotechnology, and medical devices, maintaining product purity is essential for both safety and regulatory compliance. Drug formulations, biologics, vaccines, and sterile medical products are often stored for extended periods of time in containers or delivery systems that may interact with the product.
Packaging materials and device components frequently contain complex mixtures of polymers, additives, pigments, lubricants, antioxidants, and manufacturing residues. Although these substances are present in small amounts, they may migrate into the product when exposed to solvents, temperature fluctuations, or prolonged contact.
Even trace levels of leachable compounds can potentially affect the stability, safety, or efficacy of a pharmaceutical product. Certain chemicals may interact with active pharmaceutical ingredients, alter formulation stability, or pose toxicological concerns. For this reason, regulatory authorities around the world require comprehensive extractables and leachables assessments for many pharmaceutical packaging systems and medical devices.


Designing Comprehensive Extractables Studies
Extractables studies are typically conducted during the early stages of product development to identify potential chemical compounds that could be released from packaging materials or device components. These studies are intentionally performed under exaggerated laboratory conditions designed to maximize the extraction of chemical constituents from the material.
During these experiments, materials may be exposed to various solvent systems that represent different chemical environments. These solvents are selected to mimic the potential interactions that could occur with pharmaceutical formulations, biological products, or other stored substances. Elevated temperatures, extended exposure times, and agitation may also be used to increase the likelihood of extracting potential chemical migrants.
The goal of extractables testing is not necessarily to simulate real-world storage conditions but to identify the complete range of chemical substances that could theoretically migrate from the material. Once these compounds are identified, scientists can assess which of them may pose a realistic risk under normal storage or usage conditions.
Evaluating Leachables Under Realistic Conditions
While extractables studies identify potential chemical migrants, leachables studies focus specifically on compounds that actually appear in the product during normal storage or use. These studies typically involve analyzing drug products, biological formulations, or other stored materials after they have been exposed to packaging components for defined periods of time.
Leachables testing is often performed as part of stability studies, where pharmaceutical products are stored under controlled environmental conditions for months or even years. Analytical testing is periodically conducted to detect trace levels of chemicals that may have migrated from packaging materials into the formulation.
Identifying leachables requires highly sensitive analytical instrumentation capable of detecting compounds at extremely low concentrations. Even when present in trace amounts, these substances must be carefully evaluated to determine whether they pose any potential safety concerns.
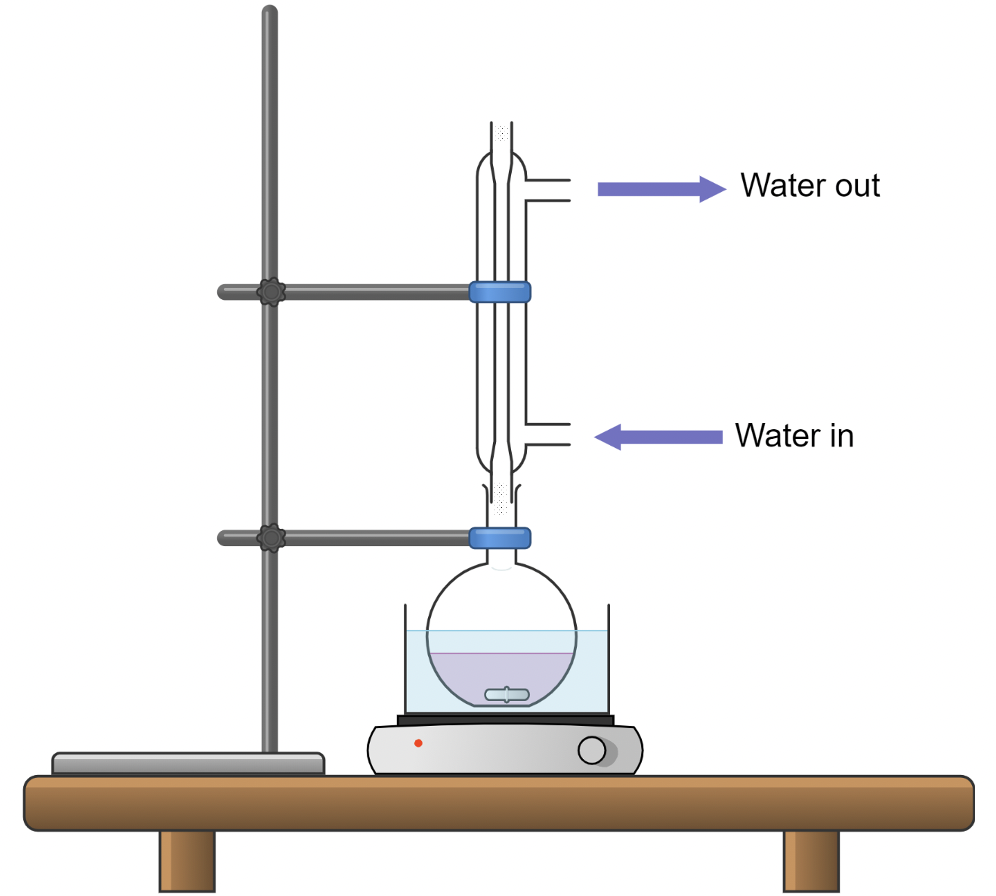
Advanced Analytical Techniques for E&L Testing
Extractables and leachables studies rely heavily on advanced analytical techniques capable of detecting and identifying trace-level chemical compounds. Because packaging materials and polymers often contain complex mixtures of additives and processing residues, multiple analytical approaches are typically required to fully characterize potential migrants.
Chromatographic methods are widely used to separate individual chemical components within complex mixtures. When combined with mass spectrometry, these techniques allow scientists to identify unknown compounds based on their molecular structure and fragmentation patterns. Spectroscopic methods further provide detailed information about chemical bonds and functional groups present in the compounds.
Elemental analysis techniques may also be used to detect inorganic substances such as metal catalysts, pigments, or stabilizers that could potentially migrate into products. In many cases, a combination of analytical methods is required to ensure that both organic and inorganic leachables are accurately identified.
Supporting Product Development and Risk Assessment
Extractables and leachables testing provides valuable information that supports multiple stages of product development. During early development, E&L studies help manufacturers select packaging materials that are compatible with their formulations and unlikely to introduce unwanted contaminants.
As products move closer to commercialization, the data generated from E&L studies becomes an important part of regulatory submissions and safety evaluations. Toxicological assessments may be performed to evaluate the potential health impact of identified compounds and determine acceptable exposure limits.
In addition to regulatory compliance, E&L testing can help manufacturers improve packaging design, select more stable materials, and reduce the likelihood of product recalls or safety concerns. By understanding how materials interact with stored products, organizations can make informed decisions that protect product quality and patient safety.


Applications Across Multiple Industries
Although extractables and leachables testing is most commonly associated with pharmaceutical and medical device industries, its importance extends to many other sectors where product purity and chemical compatibility are critical.
Biotechnology companies rely on E&L studies to evaluate materials used in bioprocessing equipment, tubing, and storage systems. Chemical manufacturers may perform similar testing to ensure that packaging materials do not introduce impurities into sensitive chemical products.
Food packaging materials may also be evaluated for potential migration of additives or contaminants into food products. In each of these industries, understanding how materials interact with stored substances helps ensure safety, quality, and regulatory compliance.
